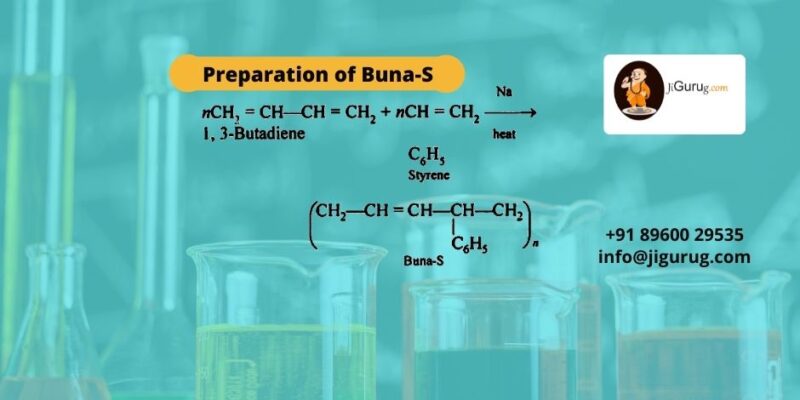
Preparation of Buna-S
If you want to know about Buna-S properties, here is everything you should understand. Buna-S is also known as Styrene-Butadiene. It is considered as the replacement for natural rubber. In other words, Buna-S is a synthetic rubber created using two monomers named Butadiene and Styrene. It is used in many industries which depend on rubber.
Buna-S – Natural Rubber
They have the effective abrasion resistance and aging stability is also more. Millions of tons of Buna-S were processed across the globe to manufacture car tires and in other industries as well. However, fifty percent of the materials are used in the automobile industry. The ratio of styrene and Butadiene can affect the properties of Buna-S. When Styrene is high, the rubber products will be hard and consist of fewer rubber properties. The polymer was invented during World War II. Walter Block, a chemist in Germany discovered Buna-S as a substitute for natural rubber.
The preparations of Buna-S indulge the emulsion polymerization of Styrene and Butadiene where Butadiene is 1/3rd ratio of Styrene. A catalyst is also used to produce cold rubber. This rubber is also known as Styrene Butadiene rubber. With sulfur vulcanization, the rubber preparation comes to an end. Though this synthetic rubber is inferior to natural rubber, its physical properties are incredible.
Important questions –
Ques: What is Buna-S?
Ques: How can Buna-S be prepared?
Ques: What are Buna-S and its uses?
Ques: What is the use of sodium in Buna-S?
Ques. What are the monomers of Buna-S rubber?
Which one of the following is an example of thermosetting polymers?
A. Neoprene B. Buna-N C. Nylon- 6,6 D. Bakelite
Which plastic is called a fire proof plastic?
A.Melamine B.Bakelite C.Teflon D.Acrylic
Caprolactum is the monomeric unit of
A) Nylon 6, 6 B) Nylon 2-Nylon-6 C) Nylon 6 D) None of these
Which of the following is not a characteristic of thermosetting polymers?
A. Linear or slightly branched long chain polymers. B. Heavily branched and cross-linked polymers. C. Become infusible on moulding. D. Cannot be remoulded or reused on heating.
Which monomer is used in the preparation of Orlon?
A. CF2=CF2 B. CH2=CH−CN C. CH2=CH−Cl D. CH2=CH−OH
